Best Practices for Managing ADC-Related Toxicity in HER+ Metastatic Cancer
HER2-directed antibody-drug conjugates (ADCs) play an important role in the treatment of breast and gastric cancer.
HER2-directed antibody-drug conjugates (ADCs) play an important role in the treatment of breast and gastric cancer. The first approval of ado-trastuzumab emtansine, also called T-DM1 (Kadcyla), put ADCs on the map for solid tumors and made the management of their adverse events (AEs) a subject of interest for oncology nurses and nurse practitioners alike.1
“The landscape of HER2-positive breast cancer has changed significantly in the past couple of years, and it’s really opened up options for our patients, which is awesome,” said Jamie Carroll, MSN, APRN, CNP, during a recent OncLive Peer Exchange®. She noted that data from DESTINY-Breast03 (NCT03529110) and other trials presented at the 2022 ASCO Annual Meeting have redefined standards of treatment for solid tumors.2 This Peer Exchange was structured to highlight realworld scenarios and put best nursing practices into context. During the discussion, oncology nurse experts examined optimal management of metastatic breast and gastric cancer with T-DM1 and fam-trastuzumab deruxtecan-nxki (Enhertu).
Jamie Carroll, MSN, APRN, CNP

Breast Cancer
T-DM1
T-DM1 was approved by the FDA in 2013 for patients with metastatic HER2-positive breast cancer who had already received treatment with taxanes, trastuzumab (Herceptin), or both.1 The approval was supported by findings from the phase 3 EMILIA trial (NCT00829166; Table), which demonstrated that T-DM1 conferred better survival than capecitabine plus lapatinib. Patients who received it achieved a progression-free survival (PFS) of 9.6 months vs 6.4 months in the control arm (HR, 0.65; 95% CI, 0.55-0.77; P < .001) and a median overall survival (OS) of 30.9 months vs 25.1 months in the control arm (HR, 0.68; 95% CI, 0.55-0.85; P < .001).3
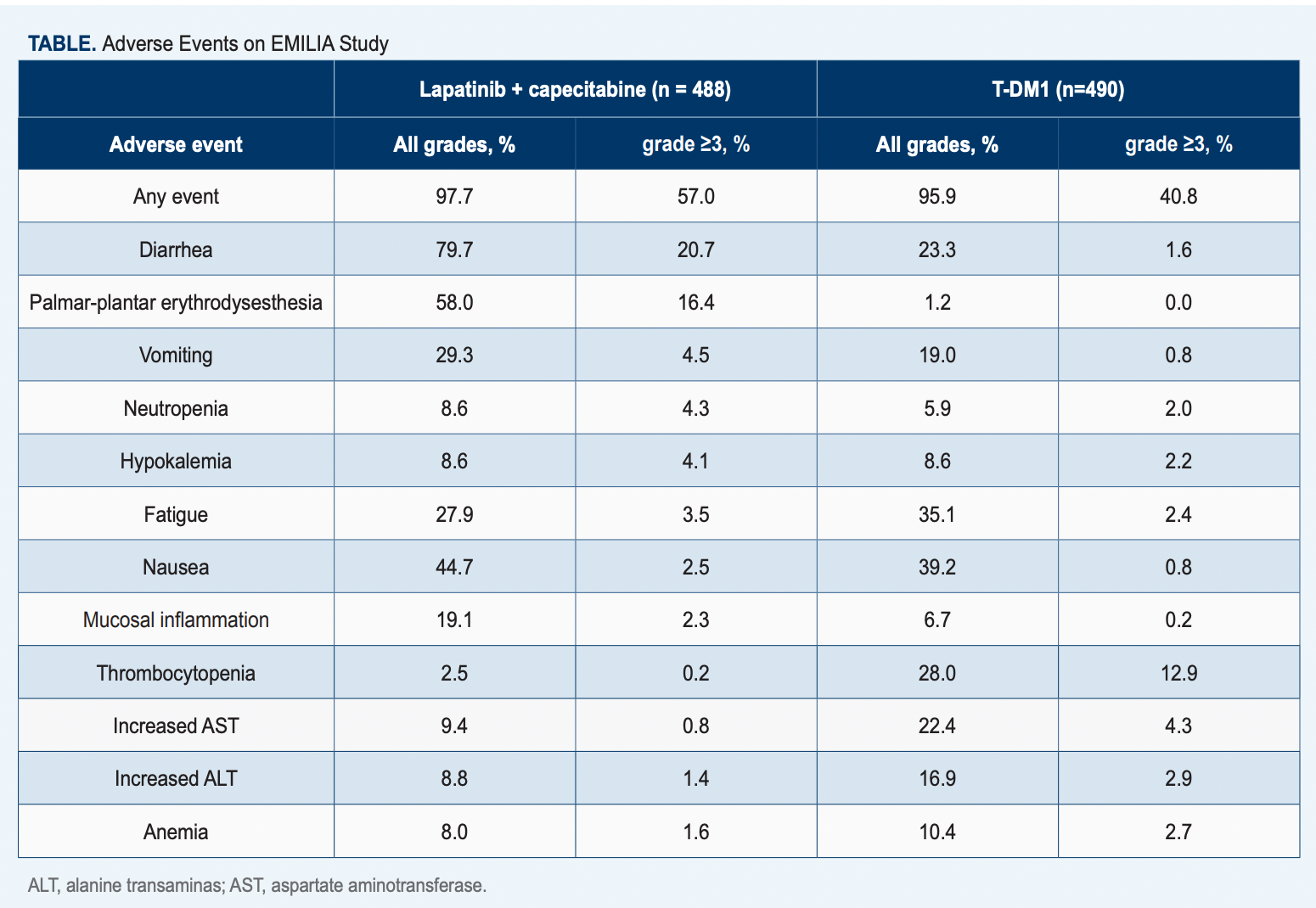
The prescribing information for T-DM1 warns about an increased risk of peripheral neuropathy, thrombocytopenia, pulmonary toxicity, and infusion-related reactions and instructs providers to monitor for signs and symptoms of neurotoxicity and temporarily withhold dose if there is grade 3 or 4 toxicity.4
Peripheral neuropathy is a fairly common AE with this treatment, said Carroll, who presented a patient case in which the AE was observed. “In the KATHERINE trial [NCT01772472], 32% of patients receiving the adjuvant treatment [with T-DM1] developed all-grade neuropathy,4 so it is important to be aware of that when caring for a patient receiving the ADC,” she noted. “You want to know their baseline.” For instance, “Do they have…contributing comorbidities such as diabetes…and peripheral neuropathy?” If they do, it “doesn’t mean that you cannot use these drugs, but it is important that you understand [patient] baseline so that you know if there [is a] progression.”

Moreover, it is critical to assess patients for this toxicity at every visit, she added. “Once [the disease is] metastatic and [patients are receiving] T-DM1, we don’t tend to see them every 3 weeks.” Carroll sees patients in cycles 1 and 2 “and then usually cycle 4, cycle 6…spacing out from there. I like to correlate visits with…echocardiograms, but I…use other members of the care team, including the chemotherapy nurses, to ask questions and let me know…of any toxicity.”
At Mayo Clinic, cryopathy is a common treatment for the symptoms of peripheral neuropathy, and gel, mittens, and booties were lent to patients every day before the pandemic. Since then, however, the practice has been discontinued owing to health concerns, but patients are encouraged to order the items online.
Trastuzumab deruxtecan
Sarah Donahue, MPH, ANP

As of October 2022, trastuzumab deruxtecan has been approved for patients with unresectable or metastatic HER2-postitive breast cancer who have previously received 2 or more anti-HER2 regimens or who have previously followed an anti-HER2 regimen in the metastatic or the neoadjuvant/adjuvant setting (with disease recurrence during or within 6 months of completing therapy) and for patients with unresectable or metastatic HER2-low breast cancer who have previously received chemotherapy in the metastatic setting or whose disease recurred during or within 6 months of completing adjuvant chemotherapy.5
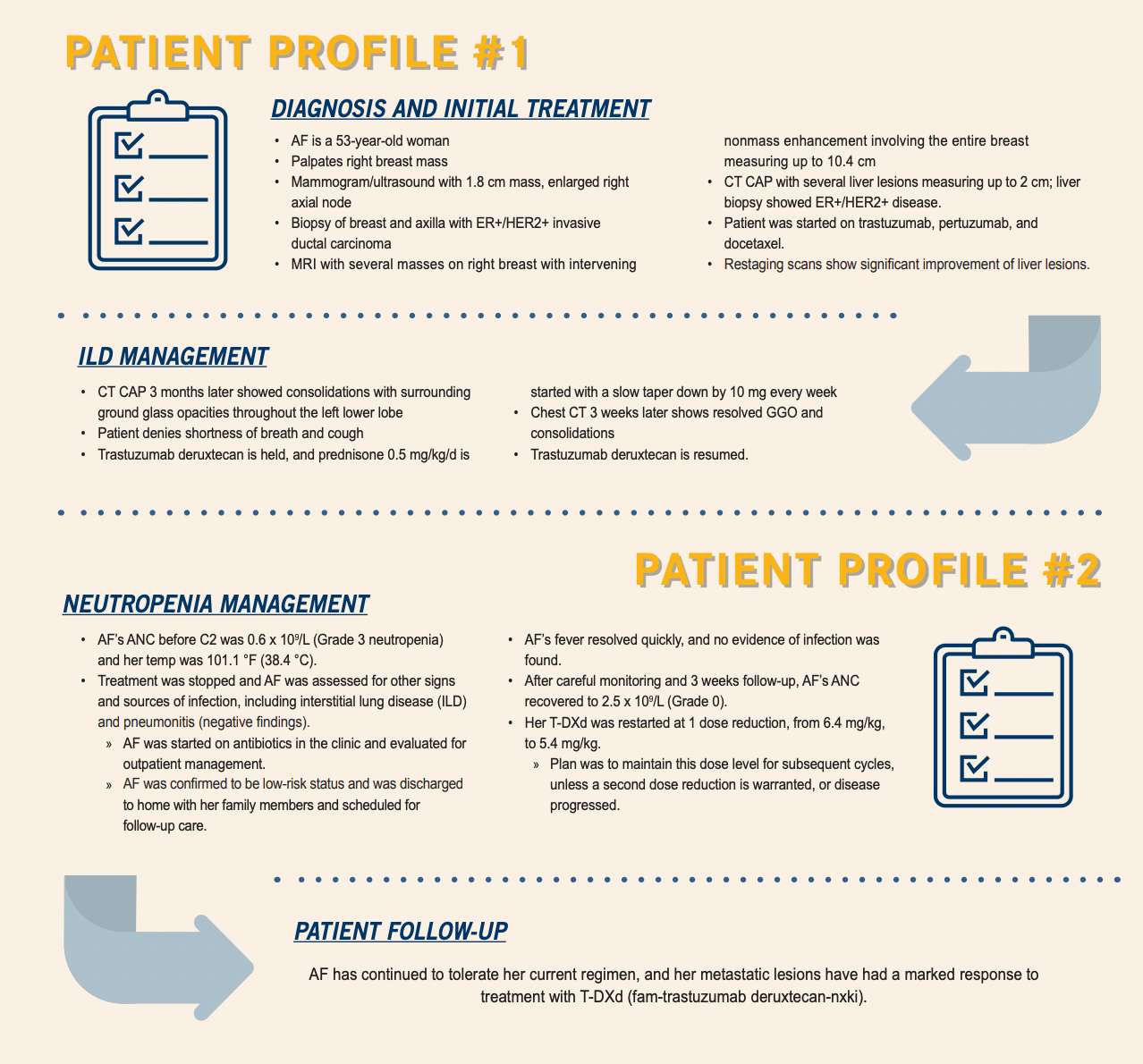
The package insert for trastuzumab deruxtecan comes with a warning for interstitial lung disease (ILD) and pneumonitis.5 However, Sarah Donahue, MPH, ANP, mentioned that in her experience these AEs are less common than reported in the DESTINY-Breast clinical trials.
“When we started giving trastuzumab deruxtecan, I felt really anxious about ILD and pneumonitis,” she said after presenting the case study of a patient who did experience ILD following treatment with trastuzumab deruxtecan (Patient Profile #1). “Any whisper of shortness of breath or cough, [and] I was ordering CT immediately,” she remarked.
According to Donahue, more recent data indicate that this AE occurs less frequently than originally thought or perhaps that its severity has been reduced through proactive intervention with steroids. Carroll agreed, saying that “even though we’re not seeing the percentages that we initially were fearful of, I still counsel my patients about letting us know if there’s any shortness of breath or any exertional dyspnea.” She observed that some patients are afraid to be honest about symptoms, thinking that they won’t be allowed to continue treatment, so it is important to encourage them not to downplay respiratory problems.
Theresa W. Gillespie, PhD, MA, FAAN

Theresa W. Gillespie, PhD, MA, FAAN, agreed the advice is excellent but pointed out that it presents a challenge for patients with metastatic disease in the lungs that is already causing such problems. They are forced to figure out whether what they’re experiencing is simply a cough or a symptom they already had and “causes [them] a great deal of anxiety.”
According to Elizabeth Prechtel Dunphy, DNP, RN, ANP, AOCN, one way to address those concerns, in addition to imaging, is to make assessment questions as unintimidating as possible. “When you’re doing your review…continue to ask about their breathing…cough, dyspnea, [but] make it less scary so that if they were to answer yes…they wouldn’t be afraid,” she advised. “Doing that on a consistent basis [eases] communication.”
Elizabeth Prechtel Dunphy, DNP, RN, ANP, AOCN

On the other hand, trastuzumab deruxtecan may not always be the best choice. “We have several options in metastatic breast cancer,” Carroll said. “If you have…a patient who has an underlying lung disease, then it might not be the best treatment right now [although it could be] in future lines.”
Clinical Pearls
According to Carroll, nurses should think about ice when it comes to ADCs. “If there’s any risk of stomatitis or peripheral neuropathy, just think ‘freeze’,” she said. “Have them chew ice in the chemotherapy unit, have them stick their hands and… feet in Elasto-Gel mittens and booties. If you’re worried about alopecia, [consider] cryotherapy to the head. ‘Just think ice’ is never wrong.” For patients experiencing dry mouth, she recommends XyliMelt, adhering discs that are placed on the gums to stimulate the salivary glands, or Jolly Ranchers, which do the same thing.
For her part, Donahue noted the importance of premedication. “When I have patients who are going to be on a regimen that’s highly immunogenic, I want to prevent [toxicity] as much as possible,” she said. “We [see] a lot of nausea, so we [administer] premedication, including… aprepitant…along with a long-acting H23 inhibitor. I give dexamethasone as a premedication, and I also make sure that they have taken-home medications such as ondansetron, lorazepam [Ativan], and prochlorperazine [Compro]. Sometimes I add…olanzapine for patients who are having a lot of nausea and often omeprazole…for the acid reflux.”
Although she is not seeing as much diarrhea as was reported in the trials, Donahue checks that patients have loperamide (Imodium) on hand at home.
Gastric Cancer
Trastuzumab deruxtecan is also a viable second-line option in metastatic gastric cancer. Its approval was supported by data from the DESTINY-Gastric01 trial (NCT03329690), in which median OS among 187 patients randomly assigned to receive targeted therapy or physicians’ choice of chemotherapy was 12.5 months in the investigational group and 8.9 months in the control (HR, 0.59; 95% CI, 0.39-0.88; P = .01).6,7 The overall response rates were 51.3% and 14.3%, respectively (P < .001).
The most commonly reported grade 3 or 4 treatment-emergent AEs in the trial were decreased neutrophil count (51% of patients) and decreased white blood cell count (21%). A small percentage of patients developed ILD, but neutropenia is of greater concern, according to Gillespie (Patient Profile #2). “In DESTINY-Gastric01, the rate of neutropenia was approximately 72%, with a considerable number [being] grade 3 or 4,” she said, whereas febrile neutropenia was reported in only 4.8% of patients.5 “Those are areas that we would definitely want to…focus on,” she added, noting that it is also important to address nausea, vomiting, and fatigue.
Myelosuppression is also common across the board in patients receiving trastuzumab deruxtecan, and certain risk factors can increase the risk of neutropenia, Gillespie continued. “Previous therapies might have left their bone marrow a little beat up,” she said. In addition, “there are lots of…conditions, including obesity, diabetes, congestive heart failure, COPD [chronic obstructive pulmonary disease], rheumatoid arthritis… osteoarthritis…[and] HIV” that suppress bone marrow function.
As with other systemic therapies that can result in neutropenia, regular monitoring of complete blood count and absolute neutrophil count—plus patient education—are critical. Unfortunately, good monitoring is still the most effective defense against neutropenia, Prechtel Dunphy said, noting that risk factors to keep in mind when prescribing are how heavily the patient has been pretreated and how compromised their bone marrow might be. In terms of education, her best advice was to involve caregivers so that they know to call the clinician if the patient develops a fever or exhibits other signs of infection.
Gillespie remarked that the only good thing about neutropenia is that it is easily detectable with a simple blood test. Nevertheless, patients must be properly taught to recognize it. “We try to emphasize that this is not something we mess around with. This is something that you report at the earliest time that it’s observed,” she said.
Fortunately, the social changes brought about by the pandemic have created a safe space for these patients to practice safe behaviors, according to Carroll. “Prior to COVID-19, if an individual was wearing a mask…in public, others would look at them, but now that’s a common thing,” she said. “If a patient is neutropenic, and we’re worried about development of neutropenic fever or dose delays, holds, anything like that, patients can wear a mask.”
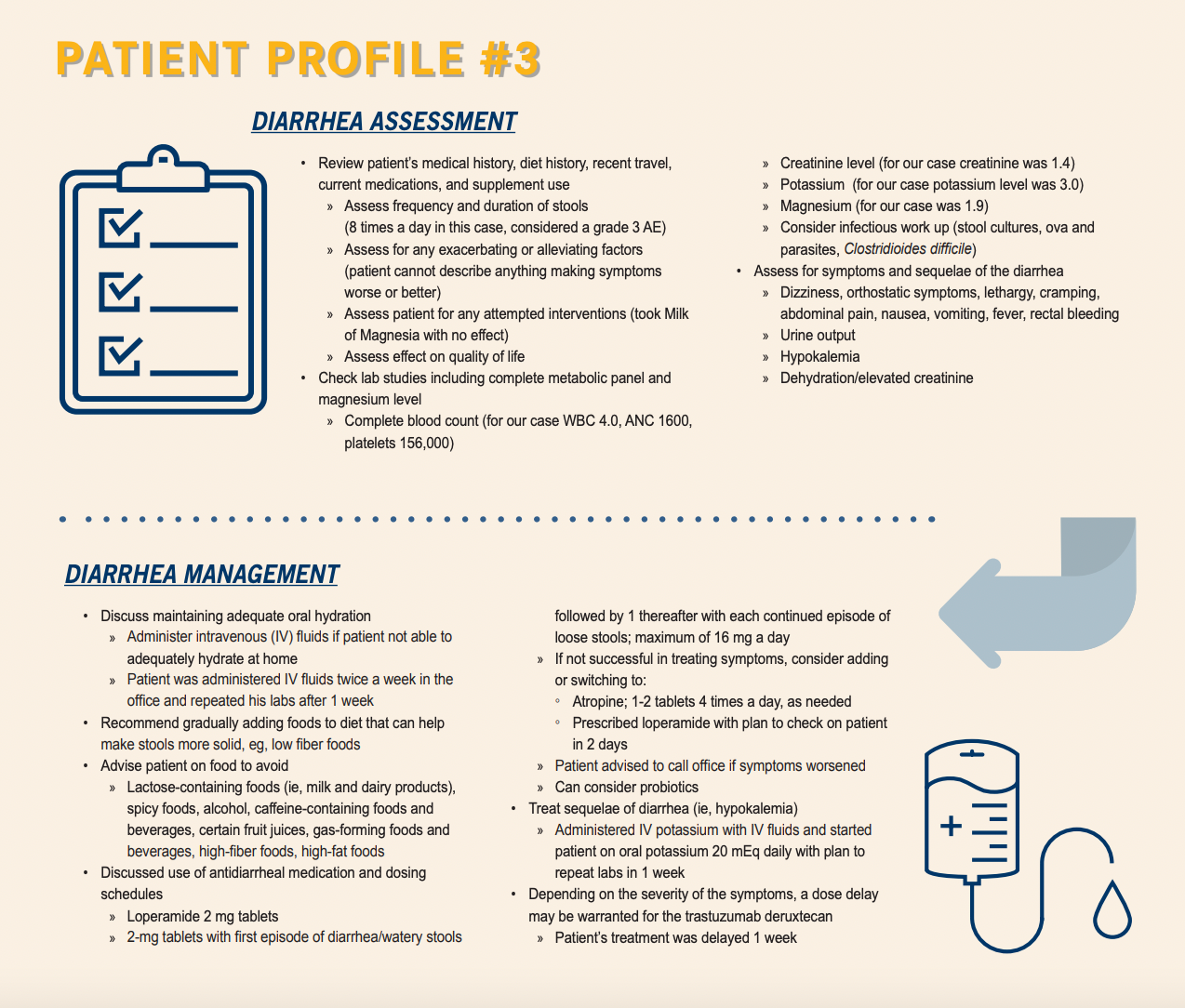
Diarrhea is another key AE in patients receiving trastuzumab deruxtecan (Patient Profile #3). In DESTINY-Gastric01, the rate of all-grade diarrhea was 32% and the incidence of grade 3 or 4 diarrhea was 2.4%.4 Prechtal Dunphy discussed her team’s management plan for a patient with diarrhea: “We talked about hydration at home, but also IV [intravenous] fluids in the office or through a home infusion company if the patient was not able to adequately hydrate at home. For [this] case, we administered IV fluids in our office twice per week [and] repeated his laboratory tests weekly.” In addition, they encouraged the patient to follow a low-fiber diet and avoid spicy and gas-producing foods, alcohol, certain fruit juices, food and beverage containing lactose, caffeine, and high amounts of fat. Antidiarrheals and probiotics were also considered.
Clinical Pearls
The best way to manage AEs in gastric cancer is with baseline assessments, Prechtal Dunphy said. “Whether you’re meeting the patient for the first time or…seeing them in follow-up or, in particular, before they start treatment, know where they are at baseline,” she said, and “involve the caregiver[s] because they are always the ‘tattletale’ in the room.” Although patients don’t always like it, she believes it’s good to have the perspective of someone who is with the patient every day. “Obviously, they can’t tell you what the patient’s feeling, but they can at least tell you what they’re observing, which…helps you make decisions.”
Gillespie agreed that baseline measurements are critical, especially when trying to diagnose a symptom. Her other message was that because high anxiety is “part and parcel” of metastatic disease, providers should be patient with their patients and encourage them to “look at it…as a marathon…rather than a sprint.” In “second- and third- and fifth-line options…there’s lots on the menu,” she said.
References
- ImmunoGen, Inc. announces FDA approval of Kadcyla (ado-trastuzumab emtansine; also known as T-DM1). News release. ImmunoGen. February 22, 2012. Accessed September 20, 2022. https://bit.ly/3LtoHBc
- Hamilton EP, Bragaia VPH, Yeo W, et al. Trastuzumab deruxtecan (T-DXd) versus trastuzumab emtansine (T-DM1) in patients (pts) with HER2-positive (HER2+) unresectable and/or metastatic breast cancer (mBC): safety follow-up of the randomized, phase 3 study DESTINY-Breast03. J Clin Oncol. 2022;40(suppl 16):1000. doi:10.1200/JCO.2022.40.16_suppl.1000
- Verma S, Miles D, Gianni L, et al; EMILIA Study Group. Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med. 2012;367(19):1783-1791. doi:10.1056/NEJMoa1209124
- Kadcycla. Prescribing information. Genentech Inc; 2022. Accessed September 7, 2022. https://bit.ly/3x0BvJm
- Enhertu. Prescribing information. Daiichi Sankyo Inc; 2022.Accessed September 7, 2022. https://bit.ly/3KWsRRL
- FDA approves fam-trastuzumab deruxtecan-nxki for HER2-positive gastric adenocarcinomas. FDA. January 15, 2021. Accessed September 7, 2022. https://bit.ly/3sxawRJ
- Shitara K, Bang YJ, Iwasa S, et al; DESTINY-Gastric01 Investigators. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N Engl J Med. 2020;382(25):2419-2430. doi:10.1056/NEJMoa2004413

Latest Conference Coverage
259 Prospect Plains Rd, Bldg H,
Monroe, NJ 08831
All rights reserved.



